 The severity of peripheral nerve dysfunction correlates with oxyhemoglobin desaturations ( Mayer et al., 1999). Consistent with functional impairment, histological studies have identified demyelination of motoneurons in resected palatal tissue in obstructive sleep apnea ( Woodson et al., 1991 Lindman and Stål, 2002 Boyd et al., 2004). Electromyographic studies of the palatopharyngeus muscle in individuals with sleep apnea show long polyphasic potentials and reduced amplitude at maximum voluntary effort ( Svanborg, 2005). Sensory nerve action potential amplitudes are reduced in individuals with obstructive sleep apnea, and treatment of sleep apnea partially reverses this defect, supporting the concept that sleep apnea contributes to this neural dysfunction ( Dziewas et al., 2007). Clinical studies have identified neural dysfunction and injury in adults with obstructive sleep apnea. In severe sleep apnea, these hypoxia/reoxygenation events may cycle every minute or two throughout sleep. Brief arousals from sleep temporarily restore patency of the airway and oxygenation. 
The pathophysiology of this syndrome involves brief sleep state-dependent obstructions of the upper airway that are associated with hypoxemia. Obstructive sleep apnea syndrome is present in >2% of the adult population and is associated with neural injury ( Young et al., 1993). It is anticipated that obstructive sleep apnea results in endoplasmic reticulum injury involving motoneurons, whereas a critical balance of phosphorylated eIF-2a should minimize motoneuronal injury in obstructive sleep apnea. Augmentation of eIF-2a phosphorylation minimizes motoneuronal injury in this model. Collectively, this work provides evidence that long-term exposure to hypoxia/reoxygenation events, modeling sleep apnea, results in significant endoplasmic reticulum injury in select upper airway motoneurons. Protection of eIF-2α phosphorylation with systemically administered salubrinal throughout hypoxia/reoxygenation exposure prevented CHOP/GADD153 activation in susceptible motoneurons. Ultrastructural analysis of rough ER in hypoglossal motoneurons revealed hypoxia/reoxygenation-induced luminal swelling and ribosomal detachment. 
In contrast, occulomotor and trigeminal motoneurons showed persistent phosphorylation of eIF-2a across hypoxia/reoxygenation, without activations of CHOP/GADD153 or either caspase. Long-term hypoxia/reoxygenation also resulted in cleavage and nuclear translocation of caspase-7 and caspase-3 in hypoglossal and facial motoneurons. Hypoxia/reoxygenation exposures across 8 weeks in adult mice upregulated the unfolded protein response as evidenced by increased phosphorylation of PERK in facial and hypoglossal motoneurons and persistent upregulation of CCAAT/enhancer-binding protein-homologous protein (CHOP)/growth arrest and DNA damage-inducible protein (GADD153) with nuclear translocation. 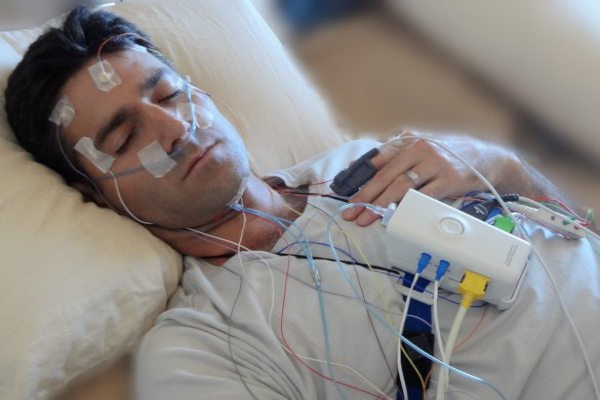 
We hypothesize that endoplasmic reticulum injury contributes to motoneuron demise. The mechanisms underlying this motoneuron injury are not understood. Hypoxia/reoxygenation exposures, modeling sleep apnea, injure select populations of neurons, including hypoglossal motoneurons. Obstructive sleep apnea is associated with neural injury and dysfunction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
